Bayer Recalls 6 mL Size Afrin Original Nasal Spray Bottles Due to Risk of Serious Injury or Illness from Child Poisoning; Violates Mandatory Standard for Child-Resistant Packaging

Description
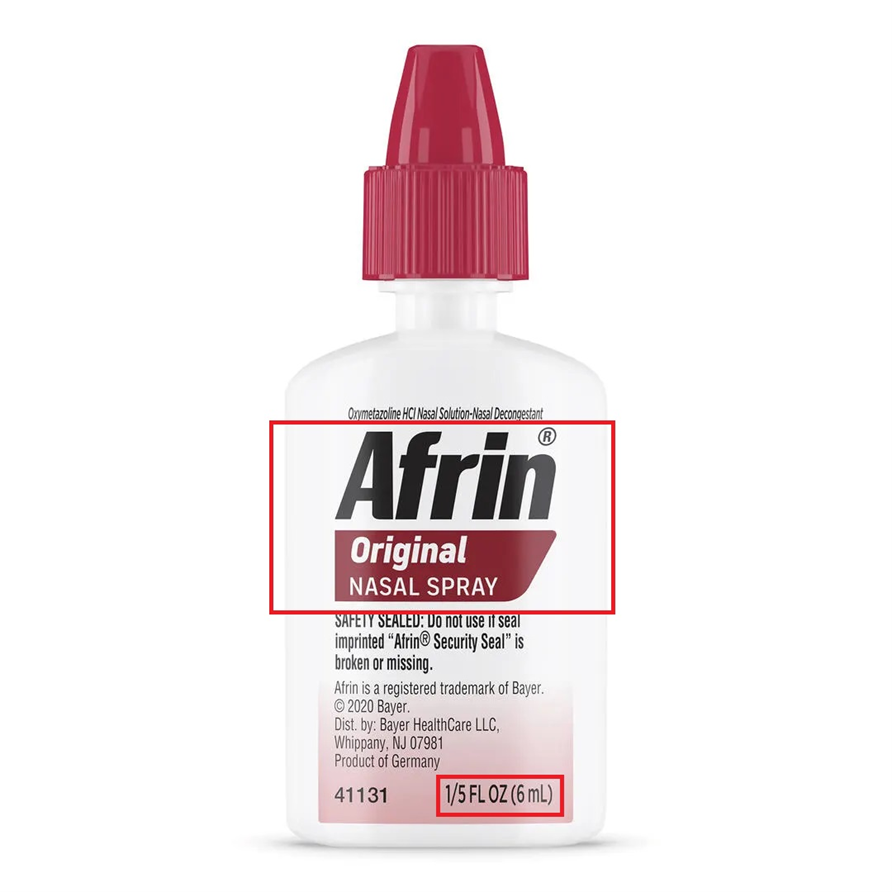
This recall involves unexpired Travel Size Afrin® Original Nasal Spray 6 mL bottles, with Lot numbers 230361, 240822, 241198, 250066, 250152, 250646, and 250831. These travel size bottles have "Afrin® Original Nasal Spray" and "1/5 FL OZ (6 mL)" printed on a label located on the front of the bottle. It is a six-digit number, followed by the expiration date in following format: "YYYYMMM". No other sizes of Afrin are included in this recall.
Hazard
The 6 mL nasal sprays contain an imidazoline, which must be in child-resistant packaging or meet the labeling requirements for non-complying packaging, as required by the Poison Prevention Packaging Act. The 6 mL nasal spray's packaging is not child-resistant nor bears the required labeling statement, posing a risk of serious injury or illness from poisoning, if the contents are swallowed by young children.
Remedy
Consumers should immediately secure the recalled bottles out of sight and reach of children and go to the webform posted on www.livewell.bayer.com/afrin-original-spray-recall to begin a request for a refund. Consumers will be asked to take and submit a photo of the product before disposing of it in order to receive a refund.